

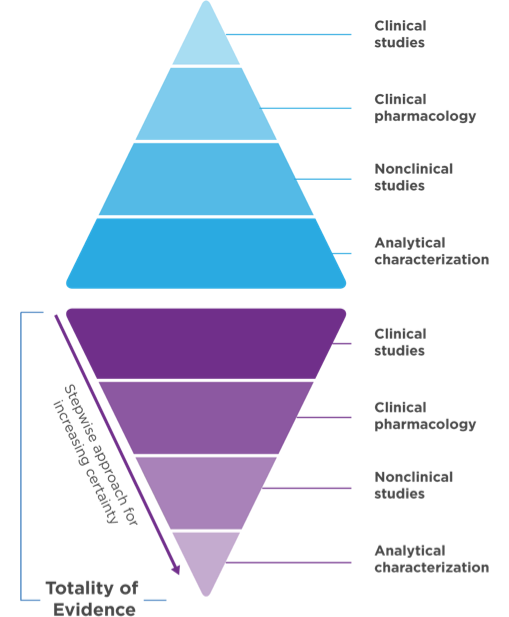
Biosimilar development1,3 Demonstrate no clinically meaningful differences in safety and effectiveness in one or more appropriate conditions of use for which the reference product is licensed. 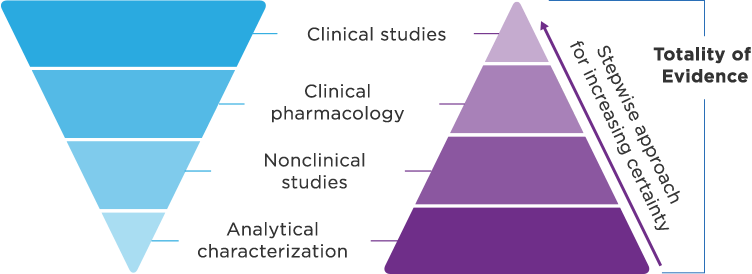

References: 1. US Food and Drug Administration. Guidance for industry: scientific considerations in demonstrating biosimilarity to a reference product. www.fda.gov/downloads/drugs/guidances/ucm291128.pdf. Accessed October 29, 2019. 2. Conner J, Wuchterl D, Lopez M, et al. The biomanufacturing of biotechnology products. In: Shimasaki C, ed. Biotechnology Entrepreneurship: Starting, Managing, and Leading Biotech Companies. Waltham, MA: Academic Press; 2014:351-385. 3. Blauvelt A, Cohen AD, Puig L, Vender R, van der Walt J, Wu JJ. Biosimilars for psoriasis: preclinical analytical assessment to determine similarity. Br J Dermatol. 2016;174:282-286. 4. Kresse GB. Biosimilars—science, status and strategic perspective. Eur J Pharm Biopharm. 2009;72:479-486. 5. European Medicines Agency. Guideline on similar biological medicinal products. www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2014/10/ WC500176768.pdf. Accessed October 29, 2019. 6. US Federal Trade Commission. Emerging health care issues: follow-on biologic drug competition. www.ftc.gov/reports/emerging-healthcare-issues-follow-biologic-drug-competition-federal-trade-commission-report/. Accessed October 29, 2019. 7. Alten R, Cronstein BN. Clinical trial development for biosimilars. Semin Arthritis Rheum. 2015;44:S2-S8. 8. European Medicines Agency. Biosimilars in the EU: information guide for healthcare professionals. www.ema.europa.edu/documents/leaflet/biosimilars-eu-information-guide-healthcare-professionals_en.pdf. Accessed October 29, 2019.